Jerusalem, Israel, June 4, 2013 / B3C newswire / - RegeneCure Ltd., a developer of novel regenerative membrane implant technology for the treatment of human and animal bone fractures, announced today that the company has started to distribute its BoneCure® Membrane product to the veterinary market around the world.
The distribution follows the successful results of a recent clinical study in several EC countries and Israel. BoneCure Membrane was used by European Veterinary Surgeons in pets for the following indications: non-union, large bone gaps, arthrodesis and severe fractures. The study showed that the healing time was about 40 percent quicker than normally observed using current Standard of Care therapies.
"Based on the outstanding safety and efficacy results of our clinical and pre-clinical studies, we expect BoneCure to enable veterinary surgeons to treat a larger number of severe fracture types and to significantly reduce the healing time and complication rate," said Moshe Tzabari, CEO of RegeneCure.
Distribution with dealers is now underway throughout Europe, Asia, Australia and the United States.
The use of the BoneCure Membrane for carpal arthrodesis is a major breakthrough in the surgery of companion animals that offers many advantages over the traditional “Gold Standard” procedure of harvesting an autogenous bone graft. This procedure, described at the BoneCure website, prevents patient morbidity and pain, reduces the risks of infection and fracture at the donor site, saves operating time and eliminates the need for a second incision.
Carpal Arthrodesis by BoneCure® Membrane
First reported case with no use of bone graft
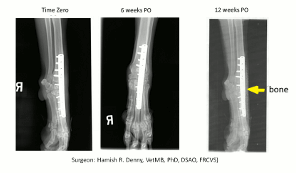
Caption: Carpal Arthrodesis treated with the BoneCure Membrane: First reported case without the use of a bone graft. For details see here.
(For high resolution picture please click the image)
Course on Small Animal Arthrodesis Sept. 27-28 in Germany
RegeneCure’s major distributor in Europe, Eickemeyer Veterinary Equipment, is organizing a practical two day course called “Small Animal Arthrodesis” on September 27 -28, 2013 at the Eickemeyer Centre for Veterinary Continuing Education, Tuttlingen, Germany. The course will be conducted by Ignacio Calvo, Ldo Vet, CertSAS, MRCVS, Head of Small Animal Orthopaedic Surgery, University of Glasgow, and Michael Farrell, BVetMed, CertVA, CertSAS, MRCVS, Senior Surgeon, Fitzpatrick Referrals. The course participants will develop theoretical and practical clinical decision-making skills about how and when to perform arthrodesis.
About BoneCure
RegeneCure's regenerative BoneCure membrane implant is cationic and has a microporous surface. Due to its osteoconductive stimulation, the membrane optimizes proliferation and differentiation of the MSCs into bone tissue in the area of need. This helps promote rapid bone healing by Guided Bone Regeneration.
The BoneCure membrane is thin and flexible, drillable and easily sutured in the treated area. It is easy to use in orthopedic surgery and requires no special surgical knowledge or preparation prior to use.
About RegeneCure
RegeneCure Ltd. was founded in July 2010 to develop, produce and market synthetic implants for bone reconstruction and fracture fusion for the orthopedic, dental and aesthetic fields. AMCA Membrane Implant technology is protected by worldwide patents. The company submitted a 510(k) application to the FDA in April 2012 for an initial trauma indication. The privately owned company has manufacturing facilities capable of meeting the high production standards required by European and United States authorities. In 2012, the company received ISO 13485:2003 certification.
Contacts
Dr. Harry Langbeheim
VP Business Development
RegeneCure Ltd.
This email address is being protected from spambots. You need JavaScript enabled to view it.
Aiz Baig BVSc, MBA, MRCVS
Eickemeyer Veterinary Equipment Ltd.
This email address is being protected from spambots. You need JavaScript enabled to view it.



